Practice Essentials
Mantle cell lymphoma (MCL) is a lymphoproliferative disorder derived from a subset of naive pregerminal center cells localized in primary follicles or in the mantle region of secondary follicles. MCL represents 2-10% of all non-Hodgkin lymphomas.
Clinical presentation
The male-to-female ratio in MCL is 3:1, and the age range at presentation is 35-85 years, with a median of 67 years. Findings on the history include the following:
Stage IV disease in 70% of patients
B symptoms, which include fever, night sweats, and weight loss, in 40% of patients
Generalized lymphadenopathy
Abdominal distention from hepatosplenomegaly
Fatigue from anemia or bulky disease
Less commonly, symptoms caused by extranodal involvement of GI tract, lungs, and CNS
Physical examination findings include the following:
Generalized lymphadenopathy in 75%
Splenomegaly in 60% (may be massive)
Hepatomegaly in 30%
Poor performance status in 20%
Less commonly, palpable masses in skin, breast, and salivary glands
Workup
Studies and procedures for diagnosing and staging MCL are as follows:
Lymph node biopsy and aspiration; aspiration alone is insufficient to establish a diagnosis
Use bone marrow aspirate/biopsy results for staging rather than diagnostic purposes
Immunophenotyping helps differentiate MCL from other small B-cell lymphomas
Torso CT scanning or PET scanning is important for initial staging and for assessing response to treatment
Blood studies may yield the following results in MCL:
CBC: Anemia and cytopenias are secondary to bone marrow infiltration; lymphocytosis of more than 4000/µL occurs in 20-40% of cases
Serum chemistry: Elevation of lactate dehydrogenase levels correlates with tumor burden
Liver function tests: Abnormal findings result from liver involvement
Beta2-microglobulin: An elevated level indicates a poor prognosis
Gamma globulin: Hypogammaglobulinemia or monoclonal gammopathy are rare findings
Coombs test: Rarely positive
On immunophenotyping, tumor cells in MCL are monoclonal B cells with the following characteristics:
Express surface immunoglobulin, immunoglobulin M, or immunoglobulin D
Are characteristically CD5+ and pan B-cell antigen positive (eg, CD19, CD20, CD22)
Lack expression of CD10 and CD23
Overexpress cyclin D1
Management
Reliably curative treatments for MCL are lacking. An inexorable pattern of progression is characteristic. Treatment selection takes into account patient age, fitness, and whether autologous stem cell transplantation (ASCT) is planned.
Standard first-line treatment consists of chemoimmunotherapy.[1] This may result in prolonged remission, but relapse is to be expected.[2] Options for second-line therapy in patients with relapsed/refractory disease include chemotherapy-free regimens with biologic targeted agents such as covalent Bruton tyrosine kinase (BTK) inhibitors, lenalidomide,venetoclax, and chimeric antigen receptor (CAR) T-cell therapy.[3, 1, 2] Because of their efficacy in the relapsed/refractory setting, biologic agents are increasingly used for first-line treatment of MCL.[1] See Treatment, below.
Epidemiology
Mantle cell lymphoma (MCL) is a type of non-Hodgkin lymphoma (NHL). The American Cancer Society estimates that in 2025 there will be 80,350 new NHL cases in the United States, representing approximately 4% of all cancer diagnoses.[7, 8] MCL is a relatively uncommon form of NHL, comprising 3-10% of NHL, with an annual incidence of 0.5 to 1 cases per 100,000 population. The exact international prevalence of MCL is difficult to estimate because of the lack of uniform classification and procedures used for diagnosis.
Surveillance, Epidemiology, and End Results (SEER) data show that the incidence of MCL per 100,000 population increased from 0.711 in 2000-2006 to 0.800 in 2007-2013 (P < 0.001), due mostly to an increased incidence in patients age 65 years and older. Relative 5-year survival rates showed a modest but significant increase over that period, most notably in patients 50-64 years of age (from 61.3% to 67.4%) and in those with stage IV disease (from 48.0% to 55.1%).[9]
Overall, Whites are at higher risk of developing NHLs than Blacks, Hispanics, or Asian Americans. The male-to-female ratio is 1.46:1, and most cases are diagnosed in patients age 65-74 years, with median age at diagnosis being 68 years.[8]
Etiology
No causative factor has been identified for mantle cell lymphoma (MCL) or for most other types of non-Hodgkin lymphoma (NHL). NHL has been associated with viral infection (Epstein-Barr virus, HIV, human T-lymphotropic virus type 1, human herpesvirus 6), environmental factors (pesticides, hair dyes), and primary and secondary immunodeficiency.
Nonrandom chromosomal and molecular rearrangements play a major role in the pathogenesis of many lymphomas. The association of t(11;14)(q13;q32) with MCL suggests a causative role; t(11;14) juxtaposes the CCDN1 gene with the immunoglobin heavy chain locus, leading to overexpression of cyclin D1.[1]
Workup
MCL is diagnosed in accordance with the WHO criteria for hematologic neoplasms and detection of cyclin D1 expression or the t(11;14) translocation along with mature B-cell proliferation. The National Comprehensive Cancer Network (NCCN) recommends the following studies to establish a diagnosis of MCL[10] :
Immunohistochemistry panel: CD20, CD3, CD5, CD10, BCL2, BCL6, cyclin D1, CD21, CD23, Ki-67, TP53
Cell surface marker analysis by flow cytometry: kappa/lambda, CD19, CD20, CD5, CD23, CD10
The following studies may be useful in select circumstances:
Fluorescence in situ hybridization (FISH) or cytogenetics for detection of t(11;14), t(14;18), CLL panel.
Cell surface marker analysis by flow cytometry: CD200
TP53 sequencing in patients with typical MCL with an expected aggressive clinical course or if upfront transplantation is anticipated
Immunohistochemistry – LEF1 is a diagnostic marker for chronic lymphocytic B-cell leukemia (CLL), so if present, it helps distinguish MCL from variant CLL; SOX11 or IGHV sequencing may be useful for determing clinically indolent MCL; may also help in diagnosis of MCL with CCND1 mutations, which promote resistance to ibrutinib
Risk stratification
The European Society for Medical Oncology (ESMO) recommends the 2008 MCL International Prognostic Index (MIPI) for risk stratification.[11] The MIPI includes the following risk factors[12] :
Age: 50-59 (1 point); 60-69 (2 points); ≥70 (3 points)
Eastern Cooperative Oncology Group (ECOG) performance status ≥2 (2 points)
Lactate dehydrogenase level (ratio to upper limit of normal): 0.67-0.99 (1 point); 1.00-1.49 (2 points); ≥1.50 (3 points)
White blood cell count (× 109/L): 6700-9999 (1 point); 10,000-14,999 (2 points); ≥15,000 (3 points)
Based on the MIPI score, patients can be categorized as follows[12] :
In addition, both the NCCN and ESMO recommend assaying Ki-67 proliferative antigen to evaluate cell proliferation. Low Ki-67 (< 30%) is associated with a more favorable prognosis; however, this finding alone is not used to guide treatment decisions.[10, 11]
The NCCN guidelines list the following as common presentations of indolent MCL (asymptomatic patients with indolent MCL may be candidates for active surveillance rather than therapy)[10] :
Laboratory studies
Blood studies may yield the following results in MCL:
Complete blood count: Anemia and cytopenias are secondary to bone marrow infiltration; lymphocytosis of more than 4000/µL occurs in 20-40% of cases
Serum chemistry: Elevation of lactate dehydrogenase levels correlates with tumor burden
Liver function tests: Abnormal findings result from liver involvement
Beta2-microglobulin: An elevated level indicates a poor prognosis
Gamma globulin: Hypogammaglobulinemia or monoclonal gammopathy are rare findings
Coombs test: Rarely positive
Computed tomography
Body CT scanning is important for initial staging and for assessing the patient's response to treatment.
Immunocytochemistry
Tumor cells are monoclonal B cells that express surface immunoglobulin, immunoglobulin M, or immunoglobulin D. Cells are characteristically CD5+ and pan B-cell antigen positive (eg, CD19, CD20, CD22) but lack expression of CD10 and CD23. Cyclin D1 is overexpressed. Immunophenotyping helps differentiate MCL from other small B-cell lymphomas (see the Table, below).[13]
Table. Differential Diagnosis of Mantle Cell Lymphoma by Immunophenotyping
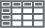
View Table | See Table |
Cytogenetics
Most cases of MCL are associated with a chromosome translocation between chromosome 11 and 14, t(11;14)(q13;q32).[3, 14]
In a study of sox11, a transcription factor involved in embryonic neurogenesis and tissue remodeling, Chen et al concluded that nuclear expression of sox11 is highly associated with MCL, but it is independent of t(11;14)(q13;q32) in non–mantle cell B-cell neoplasms.[14] Chen et al assessed expression of sox11 and evaluated its association with t(11;14) and overexpression of cyclin D1 in 211 cases of B-cell neoplasms.
The investigators noted nuclear staining of sox11 in 95% (54/57) of MCLs (98% classical and 50% variant types). Of the 3 MCLs that were negative for the nuclear sox11 staining, 2 were positive for t(11;14).[14] The remaining 114 cases of B-cell lymphomas had variable cytoplasmic positive staining without nuclear positivity.
In addition, no nuclear staining of sox11 was found in 30 plasma cell myelomas, including 12 cases with t(11;14)(q13;q32), but intense nuclear staining of sox11 was present in 50% (5/10) of a subset of hairy cell leukemias, as well as an overexpression of cyclin D1.[14] Chen et al noted that the association with cyclin D1 overexpression in hairy cell leukemia may suggest sox11 involvement in cyclin D1 upregulation in hairy cell leukemia.[14]
Diagnostic procedures
Perform lymph node biopsy and aspiration together because aspiration alone is insufficient to establish a diagnosis. Use bone marrow aspirate/biopsy results for staging rather than diagnostic purposes.
Histologic findings
In the lymph node, MCL is characterized by expansion of the mantle zone that surrounds the lymph node germinal centers by small-to-medium atypical lymphocytes. These cells have irregular and indented nuclei, moderately coarse chromatin, and scant cytoplasm, resembling smaller cells of follicular lymphoma. However, mitoses are more numerous and large cells are infrequent.
A nodular appearance may be evident from expansion of the mantle zone in 30-50% of patients early in the disease. As disease progresses, the germinal centers become effaced, with obliteration of lymph node architecture.
A blastic variant of MCL, demonstrating numerous medium-to-large blastlike cells, has been reported and is associated with a more aggressive clinical course.
In bone marrow sections, neoplastic cells may infiltrate in a focal, often paratrabecular or diffuse pattern. Diagnosis of MCL should not be based on the examination of bone marrow alone; obtaining a lymph node biopsy is required.
Treatment
Approach considerations
Standard first-line treatment for mantle cell lymphoma (MCL) consists of chemoimmunotherapy.[1] This may result in prolonged remission, but relapse is to be expected.[2] Options for relapsed/refractory disease include chemotherapy-free regimens with biologic targeted agents. Because of their efficacy in the relapsed/refractory setting, biologic agents are increasingly used for first-line treatment of MCL. Surgery is rarely indicated in MCL except for palliative procedures, such as relief of gastrointestinal obstruction.
MCL has variable behavior, from indolent to aggressive, so treatment choice in MCL is guided by risk stratification.[1] Treatment approaches based on risk stratification form the basis of various guidelines, including the widely used NCCN guideline, as discussed below
Active surveillance:
In select patients with indolent MCL—typically, those who are asymptomatic and have low tumor burden and proliferation—management can be with observation, with deferral of therapy until disease progression.[15] First-line treatment may result in prolonged remission, but relapse is to be expected.[2]
According to NCCN guidelines, active surveillance may be considered in highly selected asymptomatic patients with stage II nonbulky and noncontiguous MCL. Active surveillance may also be considered in asymptomatic patients with indolent MCL that is bulky noncontiguous stage II or stage III-IV, but in most of those cases treatment is indicated.
Involved-site radiation therapy (ISRT) with or without chemoimmunotherapy:
For stage I-II MCL that is nonbulky and noncontiguous, NCCN guidelines recommend a less-aggressive induction therapy regimen such as bendamustine plus rituximab (BR).[15] For stage I-II disease MCL that is nonbulky and contiguous, the NCCN guidelines recommend ISRT or a less-aggressive induction therapy regimen with or without ISRT.[10]
European Society for Medical Oncology (ESMO) guidelines advise that for limited nonbulky stages I-IIa MCL, shortened conventional chemotherapy induction followed by consolidation radiation therapy (RT) may be the most appropriate approach, as involved-field RT alone may not provide long-term remission. For patients with stage I-II disease who have a large tumor burden or adverse prognostic features, systemic therapy as indicated for advanced stages should be used, and consolidation RT may be considered.[11]
Treatment for patients with classical TP53 wild type
In stage II bulky noncontiguous and stage III-IV MCL and in nonfit patients with no intention for stem cell transplantation, NCCN guidelines recommend less-aggressive therapy.[10] The induction regimens include:
BR with or without acalabrutinib
VR-CAP (bortezomib [Velcade], rituximab, cyclophosphamide, doxorubicin [Adriamycin], prednisone)
R-CHOP (rituximab, cyclophosphamide, doxorubicin [hydroxydaunorubicin], vincristine, prednisone)
Lenalidomide (continuous) plus rituximab
Acalabrutinib (continuous) plus rituximab
Maintenance after less-aggressive induction therapy:
For stage II bulky noncontiguous and stage III-IV MCL in patients fit for aggressive treatment, the NCCN recommends aggressive induction therapy (see below).
Maintenance after aggressive induction therapy:
A covalent Bruton tyrosine kinase (BTK) inhibitor (eg, acalabrutinib, ibrutinib, zanubrutinib) for 2 years plus rituximab every 8 weeks for 3 years
Treatment for patients with TP53 mutations
These patients have a poor prognosis when treated with conventional therapy; the NCCN strongly recommends clinical trial enrollment in these cases[10] but otherwise recommends the folllowing regimens:
Zanubrutinib/obinutuzimab/venetoclax (in patients fit or unfit for aggressive treatment)
TRIANGLE regimen – R-CHOP plus ibrutinib, alternating with R-DHAP (rituximab, dexamethasone, high-dose cytarabine, platinum derivative [carboplatin, cisplatin, or oxaliplatin]))[16]
Less-aggressive regimens (as for wild TP53 cases)
Advanced-stage disease
For advanced-stage disease in younger patients and selected elderly fit patients, the recommended aggressive approach is induction therapy followed by consolidation therapy consisting of high-dose therapy with autologous hematopoietic stem cell transplantation) (ASCT) rescue. Prophylaxis and monitoring for tumor lysis syndrome should be strongly considered during the induction therapy.
For induction therapy, the NCCN lists the following regimens as preferred when proceeding to transplantation is intended[10] :
LyMA regimen[17] – R-DHAP followed by R-CHOP
Nordic regimen – Dose-intensified R-CHOP (maxi-CHOP) alternating with rituximab plus high-dose cytarabine
BR followed by rituximab and high-dose cytarabine
TRIANGLE regimen[16]
HyperCVAD (fractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone) plus rituximab
RBAC500 (rituximab, bendamustine, cytarabine [Ara-C] 500 mg/m2)
Modified VR-CAP/R plus cytarabine (bortezomib, rituximab, cyclophosphamide, doxorubicin, and prednisone, alternating with rituximab and high-dose cytarabine), has been used for transplant-eligible patients with MCL. A study in 37 patients reported a high rate of complete response (86%) and eligibility for autologous stem cell transplantation.[18]
In January 2025 the US Food and Drug Administration approved the use of the BTK inhibitor acalabrutinib with BR for previously untreated MCL in patients who are ineligible for autologous stem cell transplantation. Approval was based on results from the randomized controlled ECHO trial, conducted in 598 patients age 65 and older, in which median progression-free survival was 66.4 months in the acalabrutinib plus BR arm, versus 49.6 months in the placebo plus BR arm (hazard ratio 0.73, P = 0.016).[19]
For elderly patients who are not candidates for any of the above regimens, palliative chemotherapy should be considered, using milder chemo-immunotherapy regimens (eg, chlorambucil plus rituximab, BR).
For young patients (age ≤65 years), ESMO guidelines recommend induction therapy with dose-intensified immunochemotheapy (eg, R-CHOP, high-dose cytarabine) followed by ASCT with rituximab maintenance. For elderly patients, ESMO recommends conventional immunochemotherapy (eg, R-CHOP, VR-CAP, BR, R-BAC [rituximab, bendamustine, cytarabine[20] ) followed by rituximab maintenance. For compromised patients, options include R-chlorambucil, dose-reduced BR, R-CVP (rituximab, cyclophosphamide, vincristine, prednisone), or possibly best supportive care.[11]
Regimens for relapsed or refractory MCL
Biologic agents have come to dominate the treatment of relapsed or refractory MCL.[1, 2] Commonly used biologic regimens include the following[10] :
Covalent BTK inhibitors – acalabrutinib,[21] ibrutinib, zanubrutinib[22]
Non-covalent BTK inhibitor – pirtobrutinib[23]
Lenalidomide (relapsed or progressive disease after two prior therapies, one of which included bortezomib[24] ) plus rituximab
Ibrutinib plus venetoclax
Venetoclax with or without rituximab
Chemoimmunotherapeutic options, if not used in first-line therapy, include the following :
Bendamustine plus rituximab
R-BAC
Bortezomib with or without rituximab
Bortezomib, rituximab, high-dose cytarabine, and dexamethasone[25]
GemOx (gemcitabine, oxaliplatin) plus rituximab
RDHA
Salvage chemotherapy combinations followed by stem cell transplantation
Rituximab, ifosfamide, carboplatin, etoposide (R-ICE) or etoposide, methylprednisolone, high-dose cytarabine, cisplatin (ESHAP) followed by ASCT has been used.[26] However, ASCT consolidation after salvage therapy remains controversial and may benefit only a subset of patients with relapsed MCL. On the other hand, data for nonmyeloablative allogeneic stem cell transplantation are very promising, with some long-term survivors, including patients in whom prior high-dose therapy had failed.[27]
CAR T-cell therapy
In 2020, the FDA granted accelerated approval to brexucabtagene autoleucel (Tecartus), a CD19-directed genetically modified autologous T-cell immunotherapy, for the treatment of relapsed or refractory MCL in adults. Approval was based on ZUMA-2, an open-label, multicenter, single-arm, phase II trial of patients with relapsed or refractory MCL who had previously received anthracycline- or bendamustine-containing chemotherapy, an anti-CD20 antibody, and a BTK inhibitor. Of the 60 patients evaluable for efficacy, based on a minimum duration of follow-up for response of 6 months, the objective response rate was 87%, with a complete remission rate of 62%.[28, 29]
In May 2024, the FDA approved another CD19-directed CAR T-cell therapy, lisocabtagene maraleucel (Breyanzi), for adult patients with relapsed or refractory MCL based on results from an open-label, multicenter, multicohort, phase I (TRANSCEND NHL 001) trial. In 74 patients with MCL included in the primary analysis set, the objective response rate was 86.5% and the complete response rate was 74.3%. Patients had previously received 2 or more prior lines of therapy including a BTK inhibitor, alkylating agent, or CD20-targeted agent.[30]
Consultations
The following specialists may need to be consulted:
Hematologist
Oncologist
Surgeon: for lymph node biopsy, palliative procedures, and placement of a venous access device (eg, Port-a-Cath, Infus-a-Port) for chemotherapy administration
Diet
Consultation with a dietitian may be necessary for patients with poor oral intake or marked weight loss. Special attention and support is required for patients receiving chemotherapy, such as appetite stimulants or diet supplements. Patients who are neutropenic require education about food hygiene.
Follow-up
Usually, treatment and follow-up care of patients with MCL is performed in an outpatient setting. Follow-up may include the following:
Carefully monitor blood cell count prior to administering chemotherapy and 10-14 days after each treatment cycle.
Evaluate disease by monitoring history, physical examination findings, and imaging study results
Monitor adverse effects of chemotherapy by monitoring history, physical examination findings, and blood counts
Provide symptomatic treatment for adverse effects such as nausea, vomiting, diarrhea, mucositis, and fatigue
Provide psychosocial support
Admit patients for complications, disease progression, or adverse chemotherapy effects (eg, neutropenic fever or mucositis).
Complications of treatment
Complications from chemotherapy may include the following:
Infection, neutropenia, anemia, and thrombocytopenia
Fatigue
Neuropathy
Dehydration after diarrhea or vomiting
Cardiac toxicity from doxorubicin
Prognosis
Mantle cell lymphoma (MCL) is associated with a poor prognosis. It exhibits a moderately aggressive course similar to that of other types of intermediate-grade non-Hodgkin lymphoma (NHL). Unlike intermediate-grade lymphomas, it is rarely curable with currently available standard treatment.
Despite response rates of 50-70% with many regimens, MCL typically progresses after chemotherapy. Overall 5-year relative survival is approximately 50%, ranging from approximately 75% in patients younger than 50 years to approximately 36% in those age 75 years and older.[9]
The blastoid variant, commonly associated with TP53 mutations, has been associated with a worse prognosis. Gene expression profile analysis identified MCL patient subsets with more than 5 years' difference in median survival, based on cyclin D1 and other proliferation signature genes.
Using genomic and transcriptomic profiling, Li et al identified four molecular subsets of MCL that correlated with prognosis. Mutational signatures and 5-year overall survival (OS) were as follows[31] :
Mutated immunoglobulin heavy variable (IGHV), CCND1 mutation, amp(11q13), and active B cell receptor (BCR) signaling: 100% OS
del(11q)/ATM mutations and upregulation of NF-κB and DNA repair pathways: 56.7% OS
Mutations in SP140, NOTCH1, and NSD2, with downregulation of BCR signaling and MYC targets: 48.7% OS
del(17p)/TP53 mutations, del(13q), and del(9p), and active MYC pathway and hyperproliferation signatures: 14.2% OS
Author
Muhammad Rashid Abbasi, MD, Former Assistant Professor of Medicine, Albert Einstein College of Medicine; Consulting Staff, Department of Internal Medicine, Division of Hematology/Oncology, Jacobi Medical Center, Morristown Memorial Hospital, and St Clare's Hospital
Disclosure: Nothing to disclose.
Coauthor(s)
Joseph A Sparano, MD, Deputy Director, Tisch Cancer Institute, Chief, Division of Hematology/Oncology, Ezra M Greenspan, MD, Professor in Clinical Cancer Therapeutics, Icahn School of Medicine at Mount Sinai
Disclosure: Nothing to disclose.
Specialty Editors
Francisco Talavera, PharmD, PhD, Adjunct Assistant Professor, University of Nebraska Medical Center College of Pharmacy; Editor-in-Chief, Medscape Drug Reference
Disclosure: Received salary from Medscape for employment. for: Medscape.
Chief Editor
Emmanuel C Besa, MD, Professor Emeritus, Department of Medicine, Division of Hematologic Malignancies and Hematopoietic Stem Cell Transplantation, Kimmel Cancer Center, Jefferson Medical College of Thomas Jefferson University
Disclosure: Nothing to disclose.